p-V Diagrams and Cycles
Overview
A pressure-volume (p–V) diagram is one of the most important tools in thermodynamics. It shows how the pressure and volume of a gas change during a process.
From a p–V graph, you can determine:
- the thermodynamic path taken
- whether gas expands or is compressed
- work done by or on the gas
- whether the process is cyclic
- net work output of an engine
This page focuses on H2 Physics exam use of p–V diagrams.
Related hub:
Definition
A p–V diagram is a graph with pressure on the vertical axis and volume on the horizontal axis, where each point represents a thermodynamic state and each path represents a process between states.
Why It Matters
p–V graphs allow students to determine work from area, identify process type, compare different paths between same states, analyse engine cycles, and apply the first law efficiently.
Key Representations
1. Basic Structure of a p–V Diagram
- Vertical axis: pressure (P)
- Horizontal axis: volume (V)
Each point on the graph represents a unique thermodynamic state of the gas.
That state may be described by:
- pressure
- volume
- temperature
- amount of gas
For an ideal gas:
So once (P) and (V) are known (for fixed (n)), temperature is determined.
2. Meaning of a Path
A path joining two points shows how the gas changed from one state to another.
Example:
- State A → State B by one path
- State A → State B by another path
Both give the same final state, but:
- work done may differ
- heat transferred may differ
However:
- change in internal energy depends only on initial and final states
3. Work from Area Under Graph
Core Result
Work done on gas:
Therefore:
- magnitude of work = area under curve
- sign depends on direction of motion
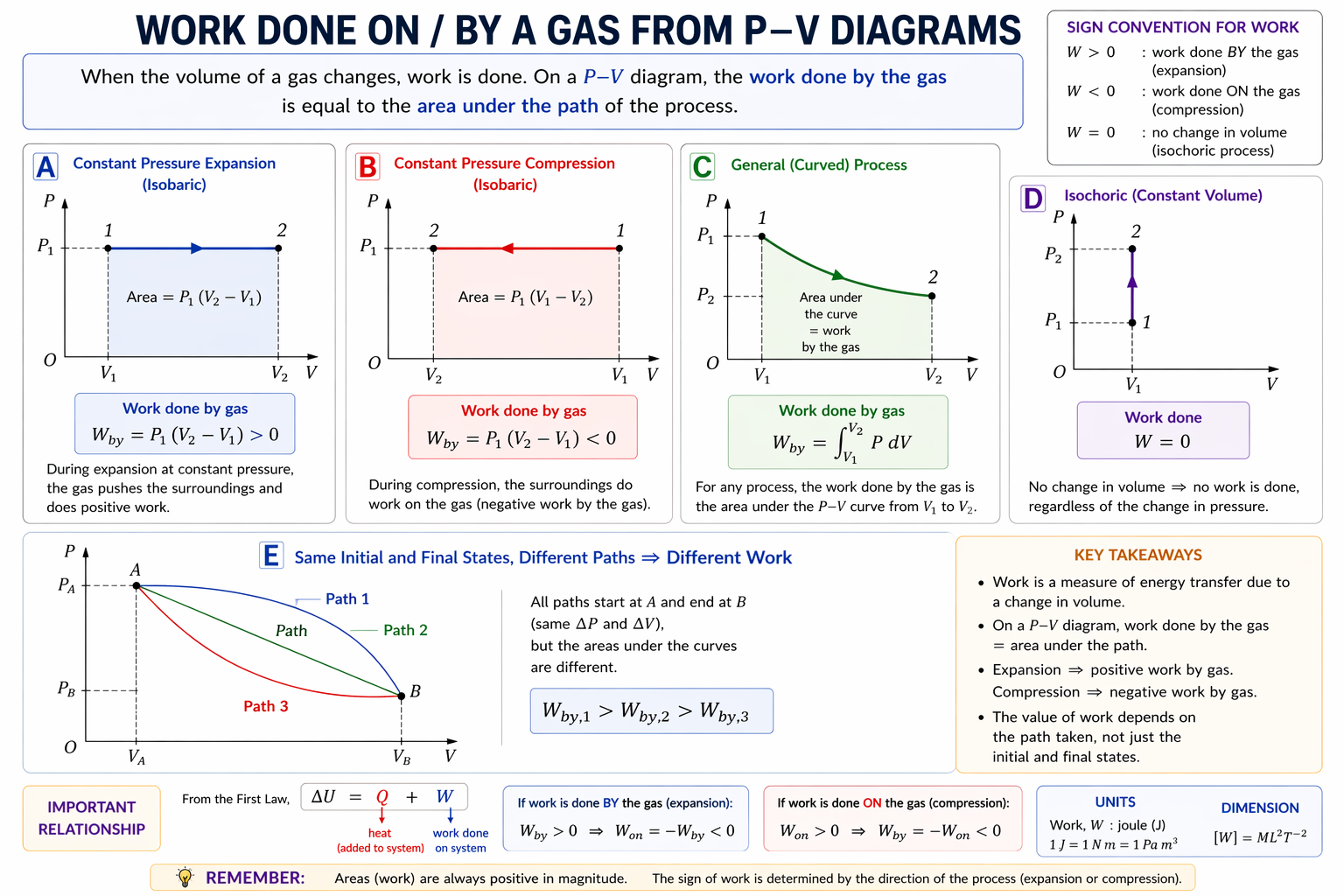
Figure: On a p-V diagram, the signed area under a path gives work done, with the sign determined by whether the gas expands or is compressed.
Always read the direction of motion along the curve before deciding the sign of the work.
4. Expansion vs Compression
Expansion
Gas volume increases (move right).
Hence:
Gas does work on surroundings.
Compression
Gas volume decreases (move left).
Hence:
Surroundings do work on gas.
5. Common Graph Shapes
5.1 Isochoric Process (Constant Volume)
Vertical line.
So:
No area under vertical path.
Interpretation
Pressure changes because temperature changes.
5.2 Isobaric Process (Constant Pressure)
Horizontal line.
Work:
Area is a rectangle.
5.3 Isothermal Process
For ideal gas:
Curve is a rectangular hyperbola.
Since:
Heat transfer balances work.
5.4 Adiabatic Process
No heat transfer:
Curve is steeper than an isothermal curve.
During expansion:
- gas cools
- pressure falls faster than in the isothermal case
6. Comparing Isothermal and Adiabatic Curves
Starting from the same point:
- adiabatic expansion curve drops more steeply
- isothermal curve falls less steeply
Reason:
- isothermal: temperature maintained
- adiabatic: temperature decreases during expansion
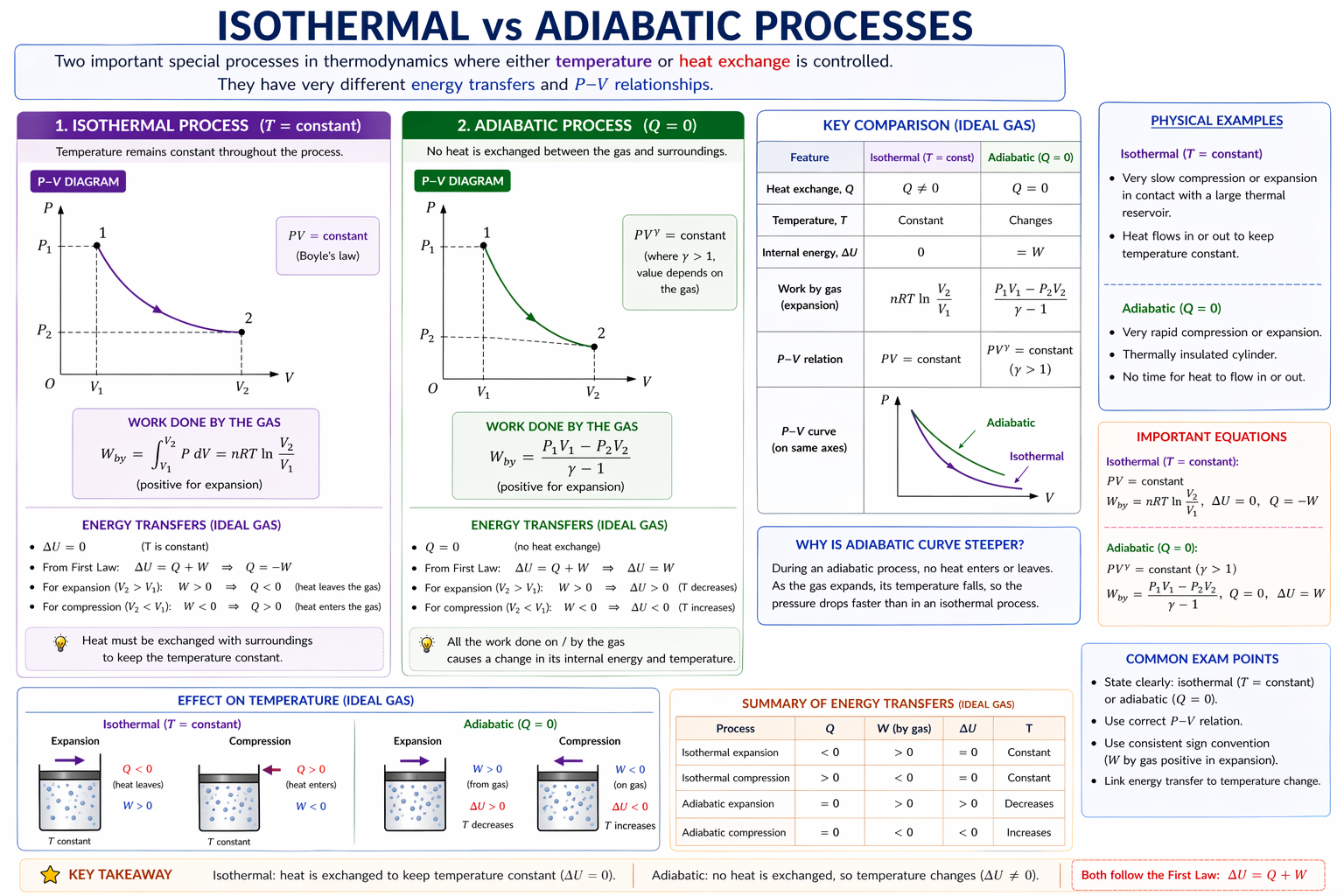
Figure: For the same initial state, an adiabatic p-V curve is steeper than an isothermal curve because no heat enters during adiabatic expansion.
This is a common comparison question: adiabatic expansion cools the gas, so pressure falls faster than in the isothermal case.
7. Cyclic Processes
A cyclic process returns gas to its original state.
Example:
A → B → C → A
Since initial state = final state:
Hence over one full cycle:
or equivalently:
net heat supplied = net work done by gas.
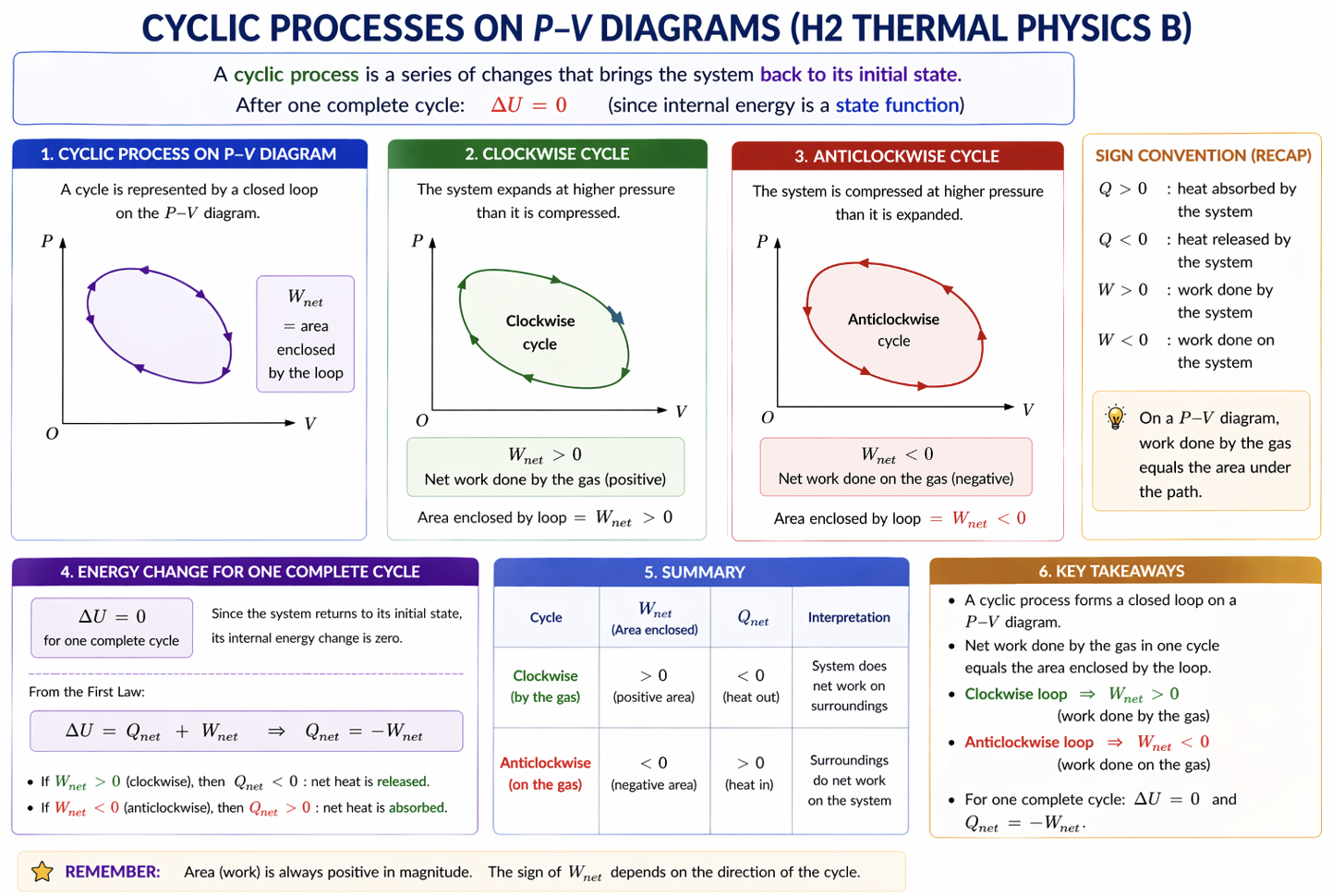
Figure: A cyclic process forms a closed loop on a p-V diagram; the enclosed area gives the net work over one full cycle.
For a full cycle, the state returns to its starting point, so (\Delta U=0) even though the net work and net heat transfer may be non-zero.
8. Enclosed Area of a Cycle
The area enclosed by a closed loop on p–V graph equals net work done in one cycle.
Clockwise Cycle
Usually:
- gas does net work on surroundings
- engine output
Anticlockwise Cycle
Usually:
- surroundings do net work on gas
- refrigerator or heat-pump style cycle
9. Engine Interpretation
A heat engine repeatedly cycles gas.
Typical idea:
- gas expands at high pressure
- gas is compressed at lower pressure
- net enclosed area = useful work output
Larger enclosed area means greater work per cycle.
10. Worked Examples
Example 1: Constant Pressure Expansion
A gas expands from:
to
at constant pressure:
Find work done on gas.
Solution
Gas does 450 J of work.
Example 2: Constant Volume Heating
Pressure rises vertically on graph.
Since:
Then:
No work done.
Example 3: Rectangular Cycle
Gas undergoes a rectangular cycle with:
- pressure difference (=2.0\times10^5,\text{Pa})
- volume difference (=4.0\times10^{-3},\text{m}^3)
Net work magnitude:
Example 4: Same Initial and Final State, Different Paths
Gas goes from A to B by two different routes.
Since internal energy is state-dependent:
is the same for both paths.
But area under graph differs, so work differs.
Hence heat transfer must also differ.
11. Reading Questions Strategically
Step 1: Identify Direction
Rightward arrow:
- expansion
Leftward arrow:
- compression
Step 2: Identify Shape
- vertical → isochoric
- horizontal → isobaric
- hyperbola → isothermal
- steeper falling curve → adiabatic
Step 3: Use Area
Find work from geometry where possible:
- rectangle
- triangle
- trapezium
Step 4: Apply First Law
12. State Property Reasoning
Internal energy of ideal gas depends on temperature only.
For monatomic ideal gas:
So if state returns to original point:
Even if large work and heat exchanges occurred during the cycle.
Links
- Thermal Physics B
- Kinetic Theory and Ideal Gases
- First Law and Thermodynamic Processes
- Thermal Physics B Common Exam Traps
Summary
p–V diagrams are not just graphs of state. They encode path, work, and cycle behaviour. Strong answers come from reading direction, recognising process shape, and using area correctly.
This equals the signed area under the curve.
Using the dossier convention:
where is work done on the system.
Expansion gives , so . Compression gives , so .
Common shapes:
| Shape | Process |
|---|---|
| Vertical line | Isochoric |
| Horizontal line | Isobaric |
| Hyperbola | Isothermal |
| Steeper falling curve | Adiabatic |
| Closed loop | Cycle |
For a cyclic process:
so:
The enclosed signed area of a closed loop gives net work done by the gas. A clockwise cycle usually represents positive net work by the gas; an anticlockwise cycle usually represents work input.
Using:
and:
for a cycle:
so:
for net values.
Common Exam Traps
Signed area under a - graph gives work done by gas, not heat.
Same start and end points do not mean same work. Work depends on path.
Closed cycle means , not zero work.
Vertical line means constant volume, not constant pressure.
Horizontal line means constant pressure, often with non-zero work.
This wiki uses , so expansion gives negative for work done on the system.