Heat Capacity and Latent Heat
Overview
This page focuses on how thermal energy changes the temperature or state of a substance.
Two major possibilities when energy is supplied:
-
Temperature changes
Use:or
-
State changes at constant temperature
Use:
This page supports:
Definition
Heat capacity describes energy needed to change temperature. Latent heat describes energy needed to change state without temperature change.
Why It Matters
This topic explains why water heats slowly, why metals heat quickly, why melting and boiling occur at constant temperature, and how calorimetry questions are really conservation-of-energy problems.
Key Representations
Heat, Temperature and Internal Energy
Heat
Heat is energy transferred due to a temperature difference.
Unit:
Temperature
Temperature measures the degree of hotness and determines direction of heat flow.
Internal Energy
Internal energy is the total microscopic energy of particles:
- random kinetic energy
- intermolecular potential energy
Supplying heat usually increases internal energy.
Heat Capacity
Definition
Heat capacity (C) of an object is the thermal energy required to raise its temperature by (1\ \mathrm{K}) (or (1^\circ\mathrm{C})).
Formula
Where:
- (Q) = thermal energy supplied
- (C) = heat capacity
- (\Delta T) = temperature rise
Unit
Notes
Heat capacity depends on:
- mass of object
- material of object
A larger object usually has a larger heat capacity.
Specific Heat Capacity
Definition
Specific heat capacity (c) is the thermal energy required to raise the temperature of 1 kg of a substance by (1\ \mathrm{K}).
Formula
Where:
- (m) = mass
- (c) = specific heat capacity
Unit
Meaning of Large or Small Specific Heat Capacity
Large (c)
Needs more energy for same temperature rise.
Examples:
- water
Implications:
- warms slowly
- cools slowly
- useful in engine cooling and climate moderation
Small (c)
Needs less energy for same temperature rise.
Examples:
- many metals
Implications:
- heats quickly
- cools quickly
Comparing Heat Capacity and Specific Heat Capacity
Heat Capacity (C)
Applies to entire object.
Specific Heat Capacity (c)
Property of material.
Relation:
Worked Example 1
A block has heat capacity:
Find heat needed to raise temperature by (15\ \mathrm{K}).
Worked Example 2
A (2.0\ \mathrm{kg}) mass of water is heated from (20^\circ\mathrm{C}) to (60^\circ\mathrm{C}).
Take:
Calorimetry and Mixing Problems
Core Principle
For an insulated system:
This follows conservation of energy.
Typical Mixing Setup
Hot object placed in cooler water.
Final temperature becomes common equilibrium temperature.
Use:
where:
- hot object cools
- cold object warms
Worked Example 3
A (0.50\ \mathrm{kg}) copper block at (100^\circ\mathrm{C}) is placed in (1.0\ \mathrm{kg}) water at (20^\circ\mathrm{C}).
Take:
- (c_{\text{Cu}}=390)
- (c_w=4200)
Find final temperature (T_f).
Water temperature changes much less because water has large thermal capacity.
Important Assumptions in Calorimetry
Usually assume:
- no heat loss to surroundings
- no evaporation
- container heat capacity negligible (unless given)
- final equilibrium reached
If calorimeter has heat capacity (C), include:
Latent Heat
Meaning
During some heating processes, temperature remains constant while energy is still absorbed.
This energy changes molecular arrangement instead of increasing average kinetic energy.
Specific Latent Heat
Formula
Where:
- (m) = mass
- (l) = specific latent heat
Unit
Types of Latent Heat
Specific Latent Heat of Fusion (l_f)
Energy required to change:
- solid (\rightarrow) liquid
at constant temperature.
Specific Latent Heat of Vaporisation (l_v)
Energy required to change:
- liquid (\rightarrow) gas
at constant temperature.
Usually:
because particles separate much more fully.
Worked Example 4
Energy to melt (0.30\ \mathrm{kg}) ice at (0^\circ\mathrm{C}):
Take:
Worked Example 5
Energy to boil away (0.50\ \mathrm{kg}) water at (100^\circ\mathrm{C}):
Take:
Heating Curves
A heating curve shows temperature against time or energy supplied.
Typical stages:
- solid warms
- melting plateau
- liquid warms
- boiling plateau
- gas warms
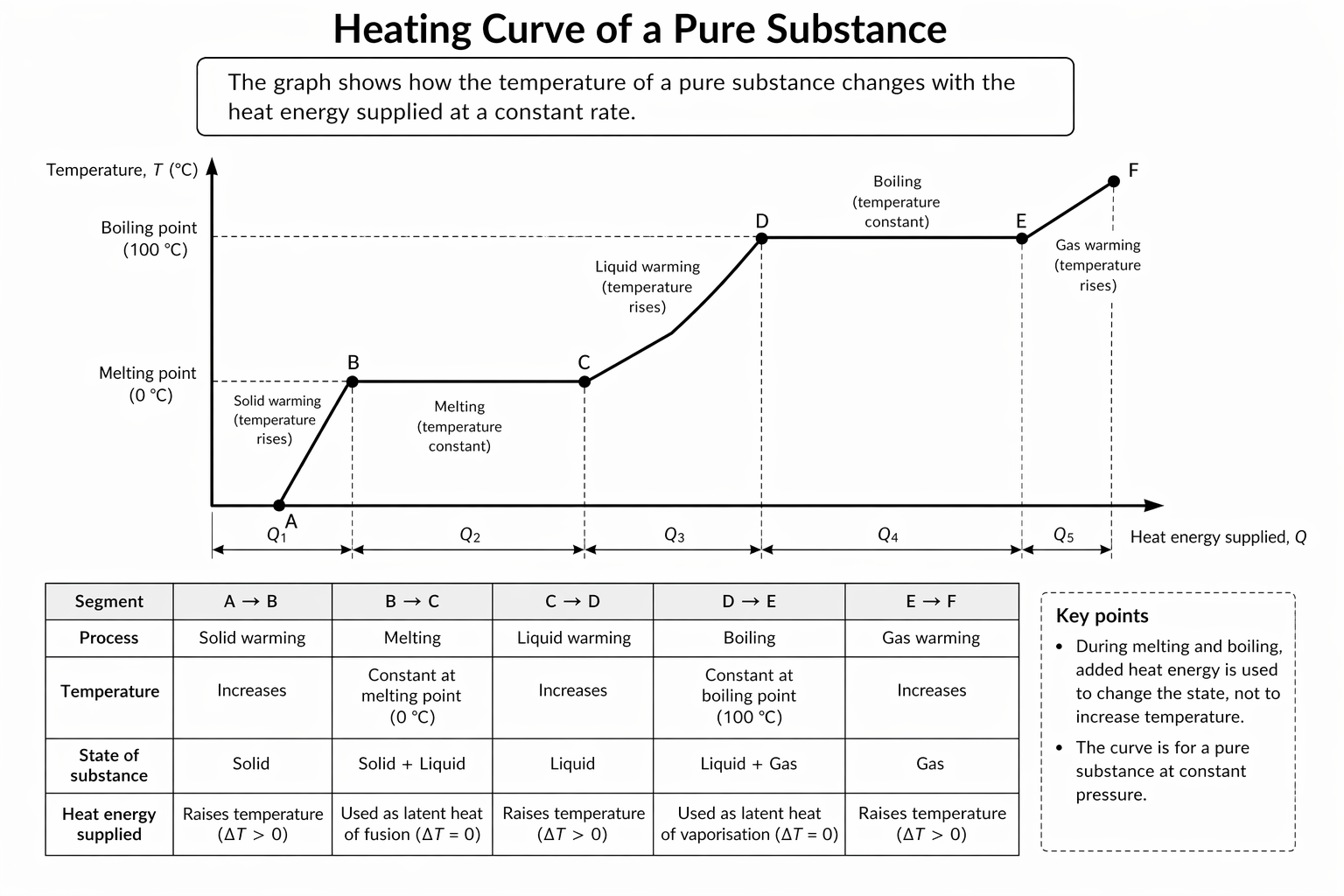
Figure: A heating curve for a pure substance, showing sloping regions for temperature rise and flat regions for phase change.
Read the graph section by section: sloping parts use (Q=mc\Delta T), while flat parts use (Q=ml).
Interpretation of Sloping Sections
Temperature rises:
Use:
Energy increases average kinetic energy.
Interpretation of Flat Sections
Temperature constant:
Use:
Energy increases separation of particles or intermolecular potential energy.
Multi-Step Energy Problems
Sometimes combine formulas.
Example: Ice at (-10^\circ\mathrm{C}) to steam at (100^\circ\mathrm{C}):
- Warm ice
- Melt ice
- Warm water
- Boil water
Use the correct equation for each stage.
Worked Example 6
Find energy to convert (0.20\ \mathrm{kg}) ice at (0^\circ\mathrm{C}) to water at (25^\circ\mathrm{C}).
Take:
- (l_f = 3.34\times10^5)
- (c_w=4200)
Step 1: Melt ice
Step 2: Heat water
Total
Links
- Thermal Physics A
- Thermal Measurement and Scales
- Thermal Practicals
- Thermal Physics A Common Exam Traps
- Work, Energy and Power
Summary
The key skill is deciding whether the energy supplied is:
- raising temperature
- changing state
- or doing both in stages
Once that is clear, the formula choice usually becomes straightforward.